Introduction
Most brightening formulas fail not because the actives are wrong but because the formulator never understood what they were actually targeting.
Tyrosinase is the copper-dependent enzyme that controls the rate-limiting step in melanin synthesis, and every credible brightening active on the market works either by inhibiting it directly, starving it of cofactors, or blocking the pathway it initiates.
Understanding the tyrosinase inhibitor category at a mechanistic level is the difference between a formula from formula chemistry that performs and one that looks good on paper.
This encyclopedia entry compares phytic acid, kojic acid, and arbutin as tyrosinase inhibitors, covering mechanism, potency, formulation behaviour, and how to combine them intelligently.
Bottom Line About Tyrosinase Inhibitor
A tyrosinase inhibitor is any compound that reduces or blocks tyrosinase enzyme activity, thereby decreasing melanin synthesis in the skin.
Phytic acid works indirectly through copper chelation, kojic acid binds the enzyme’s active copper site directly, and arbutin competes with the substrate at the same active site.
All three are cosmetically acceptable, well-documented, and suitable for leave-on formulas. Their differences in potency, stability, and pH requirements determine which one leads a formula and which ones support it.
What Is Tyrosinase: The Enzyme Behind Melanin Synthesis
Tyrosinase is a copper-containing metalloenzyme expressed in melanocytes. It catalyses two critical oxidation reactions in the melanin synthesis pathway: the hydroxylation of L-tyrosine to L-DOPA, and the subsequent oxidation of L-DOPA to dopaquinone.
Both reactions require the two copper ions housed in the enzyme’s active site to cycle between oxidation states. Without functional copper at that site, the catalytic sequence stalls and melanin production decreases at the source.
This copper dependency is precisely why chelating agents like phytic acid can function as indirect tyrosinase inhibitors. It also explains why direct active-site inhibitors like kojic acid and arbutin work at lower concentrations than chelators to achieve equivalent inhibition.
Hyperpigmentation results when tyrosinase activity is upregulated by UV exposure, inflammation, hormonal signals, or post-wound healing processes. Any formula targeting uneven skin tone must address this enzyme to achieve meaningful results.
Phytic Acid as a Tyrosinase Inhibitor
Phytic acid, INCI name Phytic Acid, CAS number 83-86-3, inhibits tyrosinase indirectly. Its six phosphate groups chelate the copper ions that tyrosinase requires for catalytic function, reducing enzyme activity by depriving it of its essential metal cofactors.
This mechanism is gentler and less targeted than direct active-site inhibition. At standard cosmetic concentrations of 0.5% to 2.0% active in leave-on formulas.
Phytic acid may help support the appearance of more even skin tone rather than delivering the rapid depigmentation results that a direct inhibitor can produce.
Its practical advantage is the four functions it delivers simultaneously. Chelation, preservation boosting, mild exfoliation, and formula stabilisation all come from one water-phase addition.
In a brightening formula, it protects other actives from metal-catalysed oxidation while contributing its own brightening mechanism through a separate pathway.
It performs best at pH 3.5 to 5.5 and should be added during cool-down below 40°C. It is incompatible with calcium-dependent thickeners and cannot serve as a standalone preservative regardless of its preservation-boosting function.
Kojic Acid as a Tyrosinase Inhibitor
Kojic acid, INCI name Kojic Acid, CAS number 501-30-4, is a direct tyrosinase inhibitor produced by fermentation of Aspergillus oryzae and related fungi.
It chelates the copper ions at the enzyme’s active site with higher specificity than phytic acid, making it a more potent brightening active at equivalent concentrations.
Its mechanism involves direct coordination with the copper ions within the catalytic site, effectively occupying the position that substrate molecules require for oxidation.
At cosmetic usage levels of 0.5% to 2.0% in leave-on formulas, its depigmentation contribution is measurable and well-documented in published literature.
The formulation challenge with kojic acid is stability. It oxidises readily in the presence of free metal ions, UV light, and elevated pH, turning the formula yellow to brown and losing efficacy as the discolouration progresses.
Pairing it with phytic acid is mechanistically intelligent because phytic acid’s chelating function removes the metal ions that catalyse kojic acid’s oxidative degradation.
It performs best at pH 3.5 to 5.0. Formulas using kojic acid benefit from chelating support, antioxidant co-actives such as ferulic acid or tocopherol, and packaging that limits light and air exposure.
Arbutin as a Tyrosinase Inhibitor
Alpha-arbutin, INCI name Alpha-Arbutin, CAS number 84380-01-8, is a glycosylated hydroquinone derivative that inhibits tyrosinase through competitive substrate inhibition.
It binds to the enzyme’s active site and competes directly with L-tyrosine for the catalytic position, slowing the hydroxylation reaction without destroying or permanently disabling the enzyme.
Alpha-arbutin is the more stable and more potent form compared to beta-arbutin. At concentrations of 0.5% to 2.0% in leave-on formulas, its tyrosinase inhibition is more targeted than phytic acid and comparable in potency to kojic acid, with a significantly better stability and tolerability profile.
Its key formulation advantage is pH flexibility. Alpha-arbutin remains stable and effective between pH 4.0 and 7.0, giving formulators considerably more working range than kojic acid allows.
It does not require the same antioxidant support infrastructure and does not discolour formulas under standard shelf conditions.
The relevant regulatory consideration is that arbutin can release trace hydroquinone under certain conditions. Under EU C
osmetics Regulation, alpha-arbutin is permitted in face products at up to 2% and body lotions at up to 0.5%. Formulators must verify current market-specific regulatory status before production, as this is factual reference information only and does not constitute legal advice.
Comparative Technical Overview
Mechanism of Inhibition
Phytic acid operates through indirect copper chelation, removing the metal cofactor tyrosinase depends on. Kojic acid binds directly to the copper ions within the enzyme’s catalytic site. Alpha-arbutin competes with L-tyrosine substrate at the active site, slowing the reaction without metal chelation.
Relative Potency
Among the three, kojic acid and alpha-arbutin deliver more targeted tyrosinase inhibition at equivalent concentrations than phytic acid. Phytic acid’s brightening contribution is real but operates alongside its other chelating functions rather than as a high-potency depigmentation agent on its own.
Stability in Formula
Phytic acid is the most inherently stable of the three and actively improves the stability of the formula it is in. Alpha-arbutin is stable across a broad pH range and requires no special protective measures at standard concentrations.
Kojic acid is the least stable, prone to oxidative discolouration, and requires chelating support, antioxidant co-actives, and light-protective packaging to maintain efficacy and appearance on shelf.
pH Working Range
Phytic acid works between pH 3.5 and 5.5. Kojic acid requires pH 3.5 to 5.0 for stability and efficacy. Alpha-arbutin offers the widest window at pH 4.0 to 7.0, making it the most formulation-flexible of the three for products targeting a near-physiological pH.
Typical Usage Levels
All three are used at 0.5% to 2.0% active in leave-on brightening formulas. These levels reflect cosmetic convention and available safety data; professional treatment formulas may exceed these ranges but require complete formula irritancy testing before production.
Combining All Three in One Formula
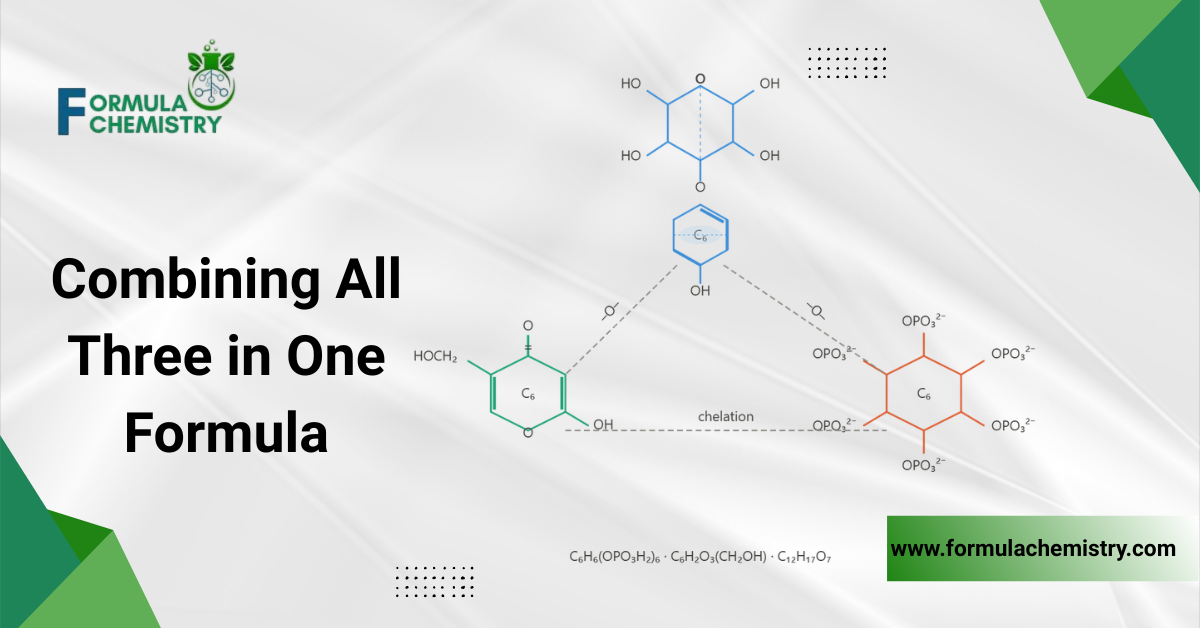
The most effective brightening formulas in this category use all three actives together rather than selecting one. Each operates through a different mechanism, and that mechanistic diversity produces additive inhibitory effect across the full tyrosinase pathway.
Alpha-arbutin carries the direct substrate competition at the active site. Kojic acid occupies the copper coordination position within the same site. Phytic acid chelates free copper ions in the broader formula environment, simultaneously starving tyrosinase of cofactors and protecting kojic acid from oxidative degradation.
A workable starting architecture places all three in the water phase at pH 3.8 to 4.5, with phytic acid added during cool-down below 40°C and kojic acid protected by ferulic acid or tocopherol acetate in the same phase. Alpha-arbutin requires no additional stabilisation at standard concentrations in this pH range.
This combination does not require the formulator to use maximum concentrations of any single active. At 1.0% active phytic acid, 1.0% kojic acid, and 1.0% alpha-arbutin, the formula delivers meaningful multi-pathway tyrosinase inhibition without overloading the formula with any one acid’s pH or irritancy contribution.
Common Formulation Mistakes
- Relying on phytic acid alone for depigmentation. Its indirect mechanism and gentler potency make it a support active, not a primary depigmentation driver. Always pair it with a direct inhibitor as the lead active.
- Using kojic acid without chelating support. Without a chelating agent in the same formula, free metal ions catalyse kojic acid’s oxidative breakdown. Phytic acid or tetrasodium EDTA must be present whenever kojic acid is included.
- Confusing beta-arbutin with alpha-arbutin in formula calculations. Beta-arbutin requires significantly higher concentrations to achieve comparable tyrosinase inhibition and carries a less favourable regulatory profile. Specifying the wrong form at the sourcing stage leads to an underperforming formula.
- Formulating the combined system above pH 5.5. Kojic acid loses stability and phytic acid loses chelating efficiency above this threshold. Even though alpha-arbutin tolerates higher pH, the limiting actives in a three-way combination set the upper pH boundary for the whole system.
- Adding kojic acid during the heated manufacturing phase. Heat and extended processing time accelerate kojic acid’s oxidative degradation before the formula even reaches the filling stage. Add it during cool-down alongside phytic acid.
- Omitting antioxidant co-actives when kojic acid is present. Tocopherol, ferulic acid, or ascorbyl glucoside in the cool-down phase extends kojic acid’s shelf stability measurably. Leaving this step out shortens the formula’s effective shelf life regardless of packaging quality.
- Exceeding regulatory concentration limits for alpha-arbutin without verification. The EU limits are 2.0% in face products and 0.5% in body products. Formulators targeting markets outside the EU must independently verify applicable limits before production.
Suitability and Safety Guidance
All three actives are suitable for normal, dry, combination, and oily skin types at standard cosmetic concentrations. Phytic acid is the most appropriate lead active for sensitive and reactive skin formulas, where its gentler indirect mechanism reduces the risk of post-inflammatory response.
Kojic acid has a moderate sensitisation profile at concentrations above 1.0% in leave-on formulas. Formulators developing products for reactive skin types should keep kojic acid at or below 0.5% and lead with alpha-arbutin or phytic acid as the primary actives.
Alpha-arbutin is well-tolerated across Fitzpatrick skin types I through VI and is a preferred option in brightening formulas for deeper skin tones where post-inflammatory hyperpigmentation is a concern. Its gentler potency relative to hydroquinone with a comparable mechanism makes it clinically appropriate in that context.
Kojic acid is not subject to restriction under the EU Cosmetics Regulation at cosmetic usage levels, but formulators should confirm current status in their specific markets. Alpha-arbutin’s concentration limits are confirmed above. All regulatory references here are factual only and do not constitute legal advice.
Always conduct a 48-hour patch test with any new formula before wider use.
Related Ingredients
Tranexamic Acid: A lysine derivative that inhibits plasmin-mediated arachidonic acid release and reduces UV-induced prostaglandin activity, working upstream of tyrosinase activation rather than at the enzyme itself. It pairs well with all three actives in this entry.
Niacinamide: Inhibits melanosome transfer from melanocytes to keratinocytes rather than suppressing melanin synthesis directly, making it mechanistically complementary to every tyrosinase inhibitor in this comparison.
Ascorbyl Glucoside: A stable vitamin C derivative that inhibits tyrosinase through copper chelation and reduces dopaquinone back to DOPA, contributing antioxidant stabilisation to kojic acid in the same formula.
FAQs About Tyrosinase Inhibitor
What is the most effective tyrosinase inhibitor?
Potency depends on mechanism and target application. Among cosmetically approved options, kojic acid and alpha-arbutin deliver more direct and measurable tyrosinase inhibition than phytic acid at equivalent concentrations.
For aggressive brightening targets, alpha-arbutin offers the better combination of potency, stability, and regulatory accessibility across major markets.
What are tyrosinase inhibitors?
Tyrosinase inhibitors are compounds that reduce or block the activity of the tyrosinase enzyme, which controls the rate-limiting step in melanin synthesis.
They work through several mechanisms including direct active-site binding, copper chelation, and substrate competition. In cosmetics, they are used to help support the appearance of more even skin tone in leave-on serums, moisturisers, and treatment formulas.
Is vitamin C a tyrosinase inhibitor?
Ascorbic acid inhibits tyrosinase through copper chelation and also reduces dopaquinone back to DOPA, interrupting the melanin synthesis pathway at two points. Its instability in aqueous formulas limits its practical use as a brightening active in most cosmetic systems.
Stable derivatives such as ascorbyl glucoside and sodium ascorbyl phosphate deliver comparable inhibitory activity with significantly better shelf stability.
How to inhibit tyrosinase in skin?
Tyrosinase inhibition in cosmetic formulas is achieved through direct active-site inhibitors like kojic acid and alpha-arbutin, indirect copper chelators like phytic acid and tetrasodium EDTA, or compounds that reduce tyrosinase upregulation signals such as tranexamic acid.
Multi mechanism approaches combining actives from different inhibitory categories consistently outperform single-active formulas. Maintaining formula pH between 3.5 and 5.5 ensures that most cosmetically approved inhibitors remain in their active forms.
What kills melanin naturally?
No cosmetic ingredient eliminates existing melanin. Cosmetic actives may help support the appearance of more even skin tone by slowing new melanin synthesis through tyrosinase inhibition, or by accelerating the natural shedding of melanin-containing surface cells through exfoliation.
Plant-derived tyrosinase inhibitors including phytic acid, kojic acid, and alpha-arbutin are the most documented options in this category within cosmetic formulation.
Does glutathione block tyrosinase?
Glutathione influences the melanin synthesis pathway by shifting production from eumelanin toward phaeomelanin, which produces a lighter pigment outcome, and by acting as an antioxidant that reduces oxidative signals that upregulate tyrosinase.
Its direct tyrosinase inhibitory activity is weaker than kojic acid or alpha-arbutin. Topical delivery of glutathione in cosmetic formulas remains a formulation challenge due to its poor skin penetration in reduced form.
What vitamins inhibit tyrosinase?
Vitamin C in its ascorbic acid form and stable derivative forms inhibits tyrosinase through copper chelation and dopaquinone reduction. Niacinamide, which is a form of vitamin B3, does not directly inhibit tyrosinase but reduces visible pigmentation by blocking melanosome transfer.
No other vitamins have well-documented direct tyrosinase inhibitory mechanisms at cosmetic concentrations in published formulation literature.
What is the strongest skin lightening agent?
What is the strongest skin lightening agent?
Hydroquinone remains the most potent and most studied tyrosinase inhibitor in dermatology, but it is a prescription or restricted ingredient in most markets and outside cosmetic formulation scope.
Among cosmetically approved actives, kojic acid and alpha-arbutin at 1.0% to 2.0% in well-formulated leave-on systems deliver the strongest tyrosinase inhibition.
A combined formula using both alongside phytic acid and a stable vitamin C derivative performs measurably better than any single active used alone.
Which is better, kojic acid or glutathione?
For tyrosinase inhibition specifically, kojic acid is the better-documented and more targeted active at equivalent concentrations. Glutathione’s brightening effect operates through a different mechanism and its topical bioavailability remains inconsistent across delivery systems.
For formulators working within cosmetic chemistry parameters, kojic acid with chelating support from phytic acid and stabilisation from ferulic acid delivers more predictable brightening performance than glutathione at standard cosmetic use levels.
Summary for Formulators
- Phytic acid inhibits tyrosinase indirectly through copper chelation and functions best as a multi-role support active, simultaneously stabilising kojic acid and contributing its own brightening pathway from a single water-phase addition.
- Kojic acid is a direct and potent tyrosinase inhibitor but demands chelating support, antioxidant co-actives, and light-protective packaging to remain stable across a standard 24-month shelf life.
- Alpha-arbutin offers direct substrate competition at the tyrosinase active site with the best stability and pH flexibility of the three, making it the most formulation-accessible direct inhibitor for most product formats.
- Combining all three at 1.0% each in a formula at pH 3.8 to 4.5 delivers multi-pathway inhibition without overloading any single acid’s contribution to irritancy or pH burden.
- No single cosmetic tyrosinase inhibitor eliminates existing pigmentation; all work by reducing the rate of new melanin synthesis, and realistic communication with clients about gradual visible improvement over consistent use is part of responsible formulation practice.
- The most common reason brightening formulas underperform is not the wrong active choice but inadequate pH control, insufficient chelating support for kojic acid, or failure to account for the 50% aqueous supply format of phytic acid in calculations.
Start by formulating a three-active brightening serum at pH 4.0 with 1.0% active phytic acid, 1.0% kojic acid, and 1.0% alpha-arbutin, then run a comparative stability study at accelerated conditions against single-active controls to observe each ingredient’s contribution to both brightening performance and formula integrity.
- What Is Phytic Acid in Skincare? Everything a Cosmetic Formulator Needs to Know
- Natural Emulsifiers for Cosmetics: HLB Values, Usage Levels & Comparison | Formula Chemistry
- Tyrosinase Inhibitors in Cosmetics: Phytic Acid, Kojic Acid, Arbutin Compared
- Phytic Acid as a Chelating Agent: How It Stabilises Your Cosmetic Formula
- Phytic Acid Serum Formula: Free DIY Cosmetic Guide with Full %

