Introduction
There is a moment every formulator recognizes standing at the bench, watching an emulsion break, wondering what went wrong. The answer is almost never the ingredient. It is almost always the chemistry behind the decision to use it.
Cosmetic chemistry is not a background subject you learn once and file away. It is the operating system running beneath every formula you build, every pH you adjust, every phase you heat and combine.
Understanding it at a working level is what separates formulators who troubleshoot confidently from those who guess and hope. This article covers the foundational science that governs cosmetic formulation the principles Formula Chemistry returns to again and again because they underpin everything else.
Why Chemistry Governs Every Formula Decision
Formulators who skip the science tend to follow recipes without understanding them. That works until it does not until a supplier substitutes one ester for another, until the pH of a new botanical extract shifts the formula outside its preservation window, until a perfectly stable bench batch fails stability testing at 40°C.
The chemistry does not change. Ingredients come and go, trends cycle, but the behavior of a lipid bilayer, the pKa of a preservative, the HLB requirement of a wax these are constants. Build your formulation thinking on those constants and the variables become manageable.
Cosmetic chemistry also intersects with regulatory compliance. The EU Cosmetics Regulation, FDA guidelines, and IFRA standards are all grounded in toxicological and chemical data. Understanding the science makes regulatory navigation significantly less opaque.
The Core Chemistry Domains Every Formulator Needs
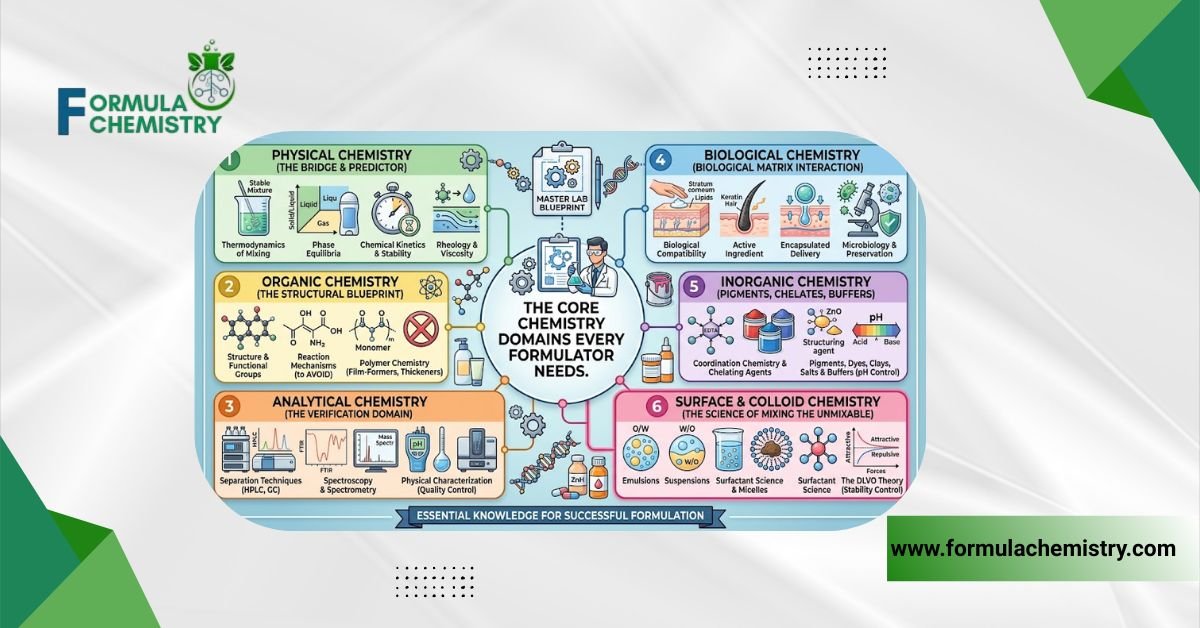
Emulsion Science
Most leave-on and rinse-off products are emulsions thermodynamically unstable systems that chemistry stabilizes kinetically. An emulsion is a dispersion of one immiscible liquid in another, held together by an emulsifier that reduces interfacial tension between the oil and water phases.
The Hydrophilic-Lipophilic Balance (HLB) system gives formulators a numerical framework for emulsifier selection. Oil-in-water emulsions generally require emulsifiers with an HLB of 8–16. Water-in-oil emulsions sit lower, typically 3–6.
When an emulsion breaks, the HLB mismatch between your emulsifier system and your oil phase is the first place to look.
Emulsifier concentration matters as much as selection. Most polymeric and fatty alcohol-based emulsifiers perform in the 2–6% range. Going under that threshold to save cost is one of the most reliable ways to fail stability testing.
Acid-Base Chemistry and pH
pH governs more in a cosmetic formula than most beginners expect. It controls:
- Preservative efficacy (most broad-spectrum systems work below pH 6.0)
- Active ingredient stability (vitamin C as L-ascorbic acid requires pH 2.5–3.5)
- Skin compatibility (the acid mantle sits at pH 4.5–5.5)
- Emulsifier performance (some self-emulsifying waxes are pH-sensitive)
A formula with the right ingredients at the wrong pH is not a good formula. Adjust with sodium hydroxide (NaOH) solution or citric acid in increments, always measuring after each addition has fully dispersed.
Preservation Chemistry
Preservation is microbiology applied to chemistry. You are creating an inhospitable environment for microbial growth, not simply adding a listed preservative and assuming the work is done.
The water activity (Aw) of your formula matters. High water activity supports microbial growth. Humectants like glycerin at concentrations above 10–15% can lower water activity and support the preservation system.
Chelating agents such as tetrasodium EDTA or phytic acid disrupt microbial cell walls and extend the efficacy of primary preservatives particularly against gram-negative bacteria like Pseudomonas aeruginosa.
Challenge testing to ISO 11930 or USP 51 is the only way to confirm a preservation system works. Bench intuition is a starting point, not a conclusion.
Rheology and Texture Chemistry
Rheology is the science of flow how a formula moves, spreads, and behaves under shear. It is controlled by:
- Carbomers (polyacrylic acid polymers) thicken water phase, activated by neutralization above pH 5.5
- Cellulose derivatives such as hydroxyethylcellulose (HEC) cold-swelling thickeners for clear gels
- Xanthan gum biopolymer that provides pseudoplastic flow, thinning under shear and recovering at rest
- Fatty alcohols such as cetyl alcohol and cetearyl alcohol contribute to emulsion viscosity and skin feel
Matching rheology modifier to formula type is not optional. A carbomer at pH 4.0 will not gel. Xanthan gum in an anhydrous formula is inert. Know your tools before you reach for them.
How Raw Material Chemistry Shapes Formula Architecture

Every ingredient you add to a formula has a chemical identity that determines its behavior. The INCI name is the entry point it tells you the compound, which tells you the chemistry.
Cetearyl Olivate (and) Sorbitan Olivate, for example, is an olive-derived, Ecocert-approved emulsifier with an HLB of approximately 8. That HLB tells you it favors oil-in-water systems and needs a co-emulsifier in heavier formulas.
The “olivate” suffix tells you it is an ester of fatty acids from olive oil, which tells you something about its skin feel and oxidative stability.
Reading an INCI name with chemical literacy is not advanced formulation. It is baseline competency, and it is what Formula Chemistry is built to develop in every formulator who works with these resources.
Active ingredients require additional chemistry knowledge. Niacinamide is water-soluble, stable across pH 4–7, and compatible with most formula systems. Retinol requires anhydrous or low-water environments and antioxidant protection. Salicylic acid requires pH below 3.5 for meaningful skin penetration. These are chemistry facts, not marketing claims.
Formulation Considerations Every Chemist Must Know
Ideal pH Range
Finished formula pH should be confirmed with a calibrated pH meter, not strips. For most leave-on skin products, target 4.5–5.5. Hair products typically sit lower, at 3.5–4.5, to keep the cuticle closed.
Solubility and Phase Addition
Every ingredient is either water-soluble, oil-soluble, or requires a dispersing agent. Adding a water-soluble active to the oil phase, or an oil-soluble antioxidant to the water phase, produces either a failed dispersion or a destabilized emulsion. Always confirm solubility before assigning a phase.
Compatibility and Incompatibilities
Cationic and anionic ingredients are chemically incompatible in most concentrations they form insoluble complexes. Cat
ionic conditioners such as behentrimonium chloride and anionic surfactants such as sodium lauryl sulfate cannot coexist at functional levels.
Divalent cations in hard water can disrupt carbomer gels and anionic surfactant systems.
Typical Usage Levels
Work within industry-established ranges. Emulsifiers: 2–6%. Carbomers: 0.3–1.0%. Xanthan gum: 0.1–0.5%. Preservatives: per supplier recommendation and regulatory limits. Exceeding usage levels wastes cost and frequently destabilizes formulas rather than improving them.
Common Mistakes and How to Fix Them
- Adding preservative at too high a temperature → Heat degrades many preservative systems, particularly phenoxyethanol-based blends → Add preservatives below 40°C, always in the cool-down phase
- Neglecting to measure final pH → A botanical extract or active shifts pH after all additions → Measure pH after every cool-down addition and adjust as needed
- Treating all emulsifiers as interchangeable → Different emulsifiers carry different HLB values and temperature sensitivities → Select based on oil phase composition and required HLB, not ingredient availability
- Skipping a chelating agent in water-heavy formulas → Tap water mineral content compromises anionic systems and creates preservation gaps → Include tetrasodium EDTA at 0.05–0.1% or a natural alternative such as gluconic acid
- Over-relying on fragrance as a preservative → Essential oils have antimicrobial properties but none meet the threshold for a compliant preservation system → Always use a validated preservative system alongside any fragrance
- Ignoring phase temperatures at combination → Adding a cool water phase to a still-hot oil phase causes thermal shock and immediate separation → Both phases should be within 5°C of each other at combination
Suitability Guide
Cosmetic chemistry principles apply across all product types and all skin and hair types. This is foundational knowledge with no exceptions. Beginners benefit most from starting with simpler systems gels, anhydrous balms, single-phase serums to observe chemistry in action before moving to emulsions where more variables interact simultaneously.
Advanced formulators working with complex actives, novel delivery systems, or regulated ingredients will find that deeper organic chemistry and toxicology knowledge becomes progressively more useful.
Always conduct a 48-hour patch test with any new formula before wider use.
FAQ’s Cosmetic Chemistry
What does cosmetic chemistry actually study?
Cosmetic chemistry studies how raw materials behave chemically and physically when combined into personal care products. It covers emulsion science, preservation, pH, and rheology. These principles govern every formula decision a chemist makes.
Why does pH matter so much in formulation?
pH controls preservative efficacy, active ingredient stability, and skin compatibility all at once. A formula at the wrong pH can fail microbiologically even with a good preservative. It also determines whether key actives remain chemically effective.
What is HLB and why should I care?
HLB is a numerical value that tells you whether an emulsifier suits oil-in-water or water-in-oil systems. Selecting an emulsifier without knowing its HLB is guesswork. Matching HLB to your oil phase is one of the fastest ways to improve emulsion stability.
Can I use tap water in my formulas?
Tap water contains minerals that interfere with anionic ingredients and preservation systems. These minerals vary batch to batch, introducing inconsistency. Deionized or distilled water below 1 µS/cm conductivity is the professional standard.
How do I know if my preservation system works?
The only reliable confirmation is a challenge test to ISO 11930 or USP 51 standards. Visual inspection and smell checks do not detect early-stage microbial growth. Send samples to an accredited lab before any formula goes to wider use.
Why do bench-stable formulas fail stability testing?
Bench conditions sit around 20–22°C, which hides weak emulsion structures and marginal preservation. Stability testing at 40°C and freeze-thaw cycling reveals those weaknesses. Treat every failure as formulation data, not a setback.
What makes two emulsifiers incompatible?
Charge conflict is the most common cause cationic and anionic emulsifiers form insoluble complexes when combined. Beyond charge, some emulsifiers compete for the oil-water interface and reduce each other’s effectiveness. Always check charge compatibility before combining emulsifier systems.
Do I need a chemistry degree to formulate well?
No, but you must understand the underlying principles that chemistry covers pH, solubility, emulsion theory, and preservation science. Many skilled formulators are entirely self-taught. What matters is whether you learn the science, not where you learned it.
Key Takeaways
- Cosmetic chemistry is the direct cause of every formulation success and failure it is not background reading, it is active knowledge
- HLB, pH, water activity, and rheology are the four mechanical systems controlling most formula outcomes
- INCI literacy is the entry point to raw material chemistry read the name, identify the compound, and its formulation behavior becomes predictable
- Preservation is a system, not a single ingredient chelation, water activity, pH, and a validated preservative all work together
- Stability testing is the only honest measure of formulation success; bench appearance is preliminary data only
Start by reviewing the pH and preservation system of every formula currently in your development pipeline those two variables account for the majority of stability and safety failures in early-stage formulas.
- Malic Acid as a Humectant in Cosmetics: The Dual Benefit Most Formulators Miss
- Cosmetic Chemistry Explained: The Science Behind Every Formula You Make
- Malic Acid: Complete Cosmetic Ingredient Profile INCI, %, pH & Formulation Notes
- Fruit Acids in Cosmetic Formulation: Malic, Tartaric, Citric Complete Guide
- Emulsifiers in Face Creams: How to Build the Right Base for AHA Formulas

